What is SOUL?
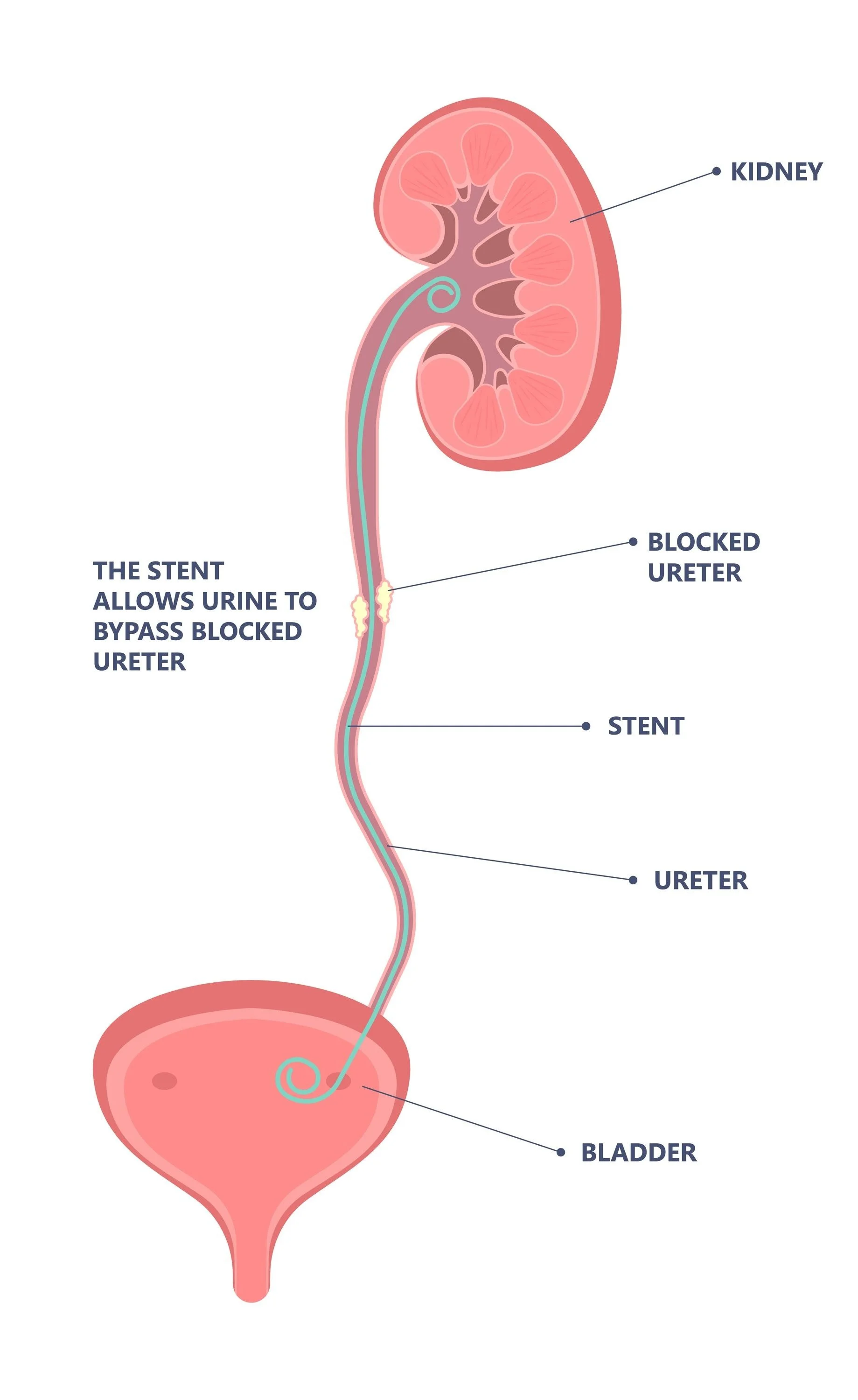
One in ten US adults will have a kidney stone in their lifetime. Ureteroscopy, an endoscopic procedure, is the most common treatment for kidney stones with >500,000 procedures performed every year. At the end of the procedure, temporary ureteral stents may be placed to help urine drain from the kidney. Many surgeons believe they prevent severe pain and emergency department (ED) visits after ureteroscopy but stents themselves are associated with pain and urinary symptoms. In fact, current US practice guidelines recommend stent omission if ureteroscopy is uncomplicated (e.g., no ureteral injury or smaller stones), which represents 90% of all cases. Despite this, stents are placed in up to 80% of all patients. The burden of stenting on patients is high. In the US,15% of patients visit the ED after ureteroscopy for symptoms, while ~50% seek ambulatory care for their pain. Many patients miss work and cannot participate in activities until the stent is removed.
A major reason for guideline discordance is the low level of evidence supporting stent omission recommendations. Studies are inconclusive on whether stents increase pain and complications. A recent Cochrane review concluded higher quality and large trials are needed to inform decision-making in this high impact area. Importantly, there is a lack of studies evaluating health-related quality of life (HRQOL), patient-reported outcomes (PROs), and unplanned healthcare utilization. Another factor is that because of experience with ureteroscopy, patients may decline to participate in randomized clinical trials (RCTs) of stents. The outcomes of these patients have been ignored in trials, limiting the
generalizability of the evidence. Combined randomized and observational studies can address this gap. Finally, surgeon and patient preferences on stenting are not characterized, leaving opportunity to inform strategies targeting practice change.
Objectives:
The objective of this clinical trial is to compare patient outcomes following Stent Omission (vs placement) after Ureteroscopy and Lithotripsy (SOUL), as previously defined using RAND Appropriateness Methodology. We will assess PROs (HRQOL, symptoms, treatment satisfaction), healthcare utilization, and factors associated with decision-making for stenting in patients undergoing ureteroscopy in the Michigan Urological Surgery Improvement Collaborative (MUSIC), comprised of diverse urology practices in Michigan and throughout the United States.
This study has the potential to improve the quality of the evidence to help patients and stakeholders. It addresses shortcomings of prior studies by: (1) Assessing PROs; (2) Assessing objective standardized healthcare outcomes; (3) Using an integrated randomized and observational trial design in a real-world setting; and (4) Improving the evidence on stent decision-making.
The trial is driven by patient partners and supported by a robust clinical registry and PRO system, allowing unprecedented efficiency to conduct clinical trials in a real-world setting. The combined randomized and observational trial design assesses outcomes and preferences in all patients. We hypothesize that patients undergoing stent omission will have improvements in PROs and 30-day healthcare utilization. There are three specific aims:
Aim 1. Compare the effectiveness of stent omission vs. placement following uncomplicated ureteroscopy. We will conduct a multi-center RCT of 264 patients undergoing ureteroscopy to compare the following: 1a. Co-Primary outcomes: PROMIS Pain Interference scores at day 7-10, 30-day healthcare Utilization; 1b. Secondary outcomes: PROs (PROMIS Pain Intensity, LURN-S10 Urinary Symptoms, ICIQ-S Satisfaction of Treatment).
Aim 2. Perform a real-world assessment of outcomes in non-randomized patients undergoing stent placement vs omission. In a separate observational cohort of patients who decline randomization and undergo uncomplicated ureteroscopy (approximately 528 patients), we will assess HRQOL, PROs and healthcare utilization in routine practice.
Aim 3. Understand decisions and preferences around stent placement using a concurrent mixed methods approach. Using interviews and surveys we will assess patient and surgeon attitudes regarding stenting. The work will inform strategies to improve the patient experience, and future study on decision-making.
Design:
The SOUL study is a pragmatic multi-center combined concurrent randomized and observational
clinical trial. The RCT arm will be stratified and randomized 1:1 to stent omission vs. placement. Stent omission vs. placement in the observation arm will be decided by the surgeon and/or patient per routine clinical practice.
All patients will be prospectively followed for 60 days after the procedure, including a variety of PRO measures administered at prespecified preoperative and postoperative timepoints. The RCT and observational components have been assessed on the Pragmatic Explanatory Continuum Indicator Summary (PRECIS 2.0) with high overall scores, indicating applicability of the intervention to usual care across a range of settings.
Sample Size and Study Duration:
The primary endpoints are the change from baseline to day 7-10 after ureteroscopy in PROMIS® Pain Interference scores and unplanned Healthcare Utilization within 30 days of ureteroscopy. To assess the co-primary endpoints, we will split the Type I error (alpha) of 5% and allocate 2.5% to each co-primary out-come (PROMIS Pain Interference & Healthcare Utilization). The rationale is that we do not think it is necessary to have both outcomes result in statistical significance to have a positive trial. If either endpoint (Reduction in Pain Interference or Healthcare Utilization) were determined to be statistically significant, then this would be sufficient to result in practice change. Eligible participants who have an uncomplicated ureteroscopy will be randomized 1:1 to stent omission vs. stent placement arms using a stratified permuted block randomization.
We will prospectively recruit adult patients >18 years undergoing uncomplicated ureteroscopy and lithotripsy at community and academic centers in MUSIC, including several centers outside Michigan. The total number of patients we need for the total study period is 792. The study duration for enrollment of patients is 2 years.
Subject Population:
Key inclusion criteria: 1) Patients ≥18 years old who have access to email or telephone; 2) Able to read and comprehend English in order to complete PROs; 3) Meet criteria for unilateral uncomplicated ureteroscopy and stent omission: largest stone size ≤ 1cm (kidney or ureter, multiple locations allowed), no use of ureteral sheath or dilation, not pre-stented
Key exclusion criteria: 1) Pregnant patients; 2) Use of ureteral access sheath or ureteral dilation; 3) Anatomical abnormalities (e.g. solitary, horseshoe, fused crossed ectopia, pelvic kidney, urinary diversion); 4) Planned secondary ureteroscopy; 5) Indwelling ureteral stent prior to ureteroscopy (pre-stented); 6) Indwelling nephrostomy tube; 7) Bilateral ureteroscopy; 8) Comorbid pain syndromes that would confound outcome assessments